Adiabatic cooling vs Evaporative cooling
Understanding the Difference Between Adiabatic Cooling and Evaporative Cooling
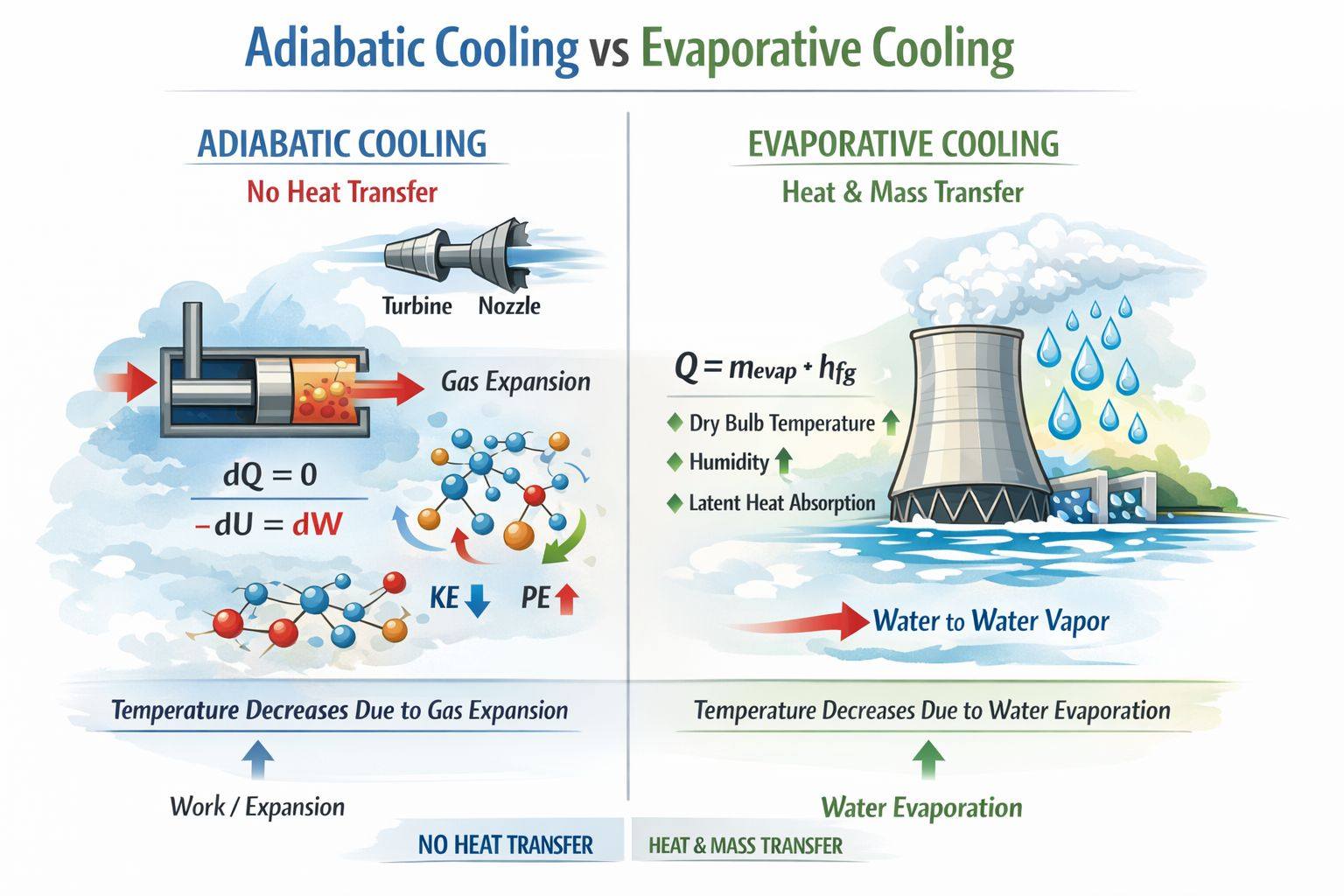
In HVAC discussions, the terms adiabatic cooling and evaporative cooling are sometimes used interchangeably, but they are actually two distinct thermodynamic processes. Let’s break down the differences.
Adiabatic Cooling
Adiabatic cooling is a process where the temperature of a system decreases without any heat transfer across its boundary.
From a thermodynamic perspective:
According to the First Law of Thermodynamics:
Since , this simplifies to:
Where:
-
= internal energy
-
= heat transfer
-
= work done by the system
In simple terms, the system cools because it performs work on its surroundings, reducing its internal energy.
From a molecular standpoint, when a gas expands, the molecules spread apart. Some of their kinetic energy (movement) is converted into potential energy (energy associated with separation). As the average kinetic energy decreases, the temperature drops—even though no heat is exchanged with the environment.
Common examples of adiabatic cooling:
-
Gas expansion through nozzles or turbines
-
Rapid expansion of gases in atmospheric processes
Evaporative Cooling
Evaporative cooling works differently—it involves both heat and mass transfer. Here, water evaporates into the air, absorbing energy (latent heat of vaporization) from the surrounding air and cooling it.
The heat removed from the air can be calculated as:
Where:
-
= evaporation rate (kg/s)
-
= latent heat of vaporization (kJ/kg)
During evaporative cooling:
-
Dry-bulb temperature decreases
-
Humidity ratio increases
-
Latent heat of the air-water system increases
Common applications of evaporative cooling:
-
Cooling towers
-
Evaporative air coolers
-
Spray-assisted condensers and dry coolers
Key Difference in Simple Terms
-
Adiabatic Cooling: Temperature drops due to expansion/work, with no heat transfer.
-
Evaporative Cooling: Temperature drops due to latent heat absorption during water evaporation, involving both heat and mass transfer.
In HVAC psychrometrics, evaporative cooling is often approximated as a constant enthalpy process. This approximation sometimes leads people to loosely call it “adiabatic,” but strictly speaking, it is not a purely adiabatic process.